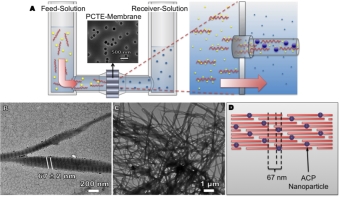
Nanodiamonds
Nanodiamonds are a fascinating material that only recently came into the spotlight despite being known for over sixty years. Recent breakthroughs in the last
decade regarding processing, purifying, and dispersing nanodiamonds enabled widespread research on diamond nanoparticles and quickly showed the inherent potential of this newly
rediscovered nanomaterial. Among the outstanding properties of the sp3 hybridized carbon nanoparticles are inherent fluorescence, excellent biocompatibility and ease of surface
functionalization. Moreover, pure detonation nanodiamonds are easy to procure without being prohibitively expensive. Based on these features, nanodiamonds are starting to be
intensely investigated as promising candidates for biomedical applications like drug delivery, nanoparticle-assisted diagnostics and imaging, or as implant coatings and
reinforcements. Nanodiamonds have generally been considered biocompatible for a broad variety of eukaryotic cells.
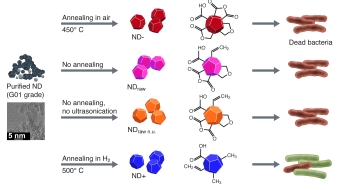
In recent work, we showed that, depending on their surface composition, nanodiamonds kill Gram-positive and -negative bacteria rapidly and efficiently. We
investigated six different types of nanodiamonds exhibiting diverse oxygen-containing surface groups that were created using standard
pretreatment methods for forming nanodiamond dispersions. Our experiments suggest that the antibacterial activity of nanodiamond is linked to the
presence of partially oxidized and negatively charged surfaces, specifically those containing acid anhydride groups. Furthermore,
proteins were found to control the bactericidal properties of nanodiamonds by covering these surface groups, which explains the
previously reported biocompatibility of nanodiamonds. We are currently starting to collaborate with several institutes to further elucidate the
antibacterial surface properties of nanodiamonds. These collaborations will enable the detailed analysis of the surface chemistry of
antibacterial nanodiamonds using XPS, Raman and NMR, as well as computational methods that model the surface/biomolecule interactions of
nanodiamond particles. In another project, we are incorporating nanodiamonds into bone replacement materials based on hydroxyapatite in
order to provide these biomaterials with antibacterial properties.